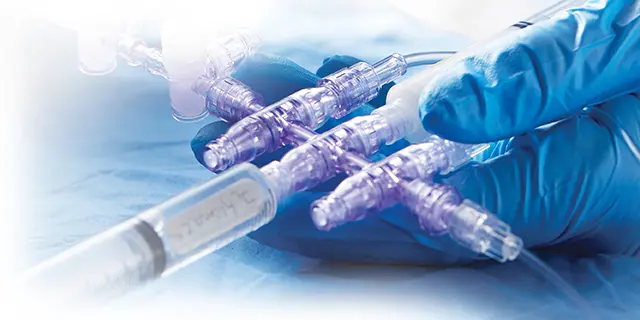
NanoClave™
Manifolds and Stopcocks
Needlefree manifolds and stopcocks featuring clinically differentiated Clave™ infection control technology
- Help reduce infection risks
- Standardize clinical protocols
- Optimize supply of IV consumables
Help to reduce infection risks while efficiently delivering IV medications

Accessing your patient’s IV line through the hub of an open stopcock or manifold may increase the risk of bacterial contamination1
While IV therapy is essential to patient care, accessing your patients' bloodstream may increase the risk of infection. As a result, the design of needlefree stopcocks and manifolds can be an important part of your efforts to minimize contamination and the risk of bloodstream infection.
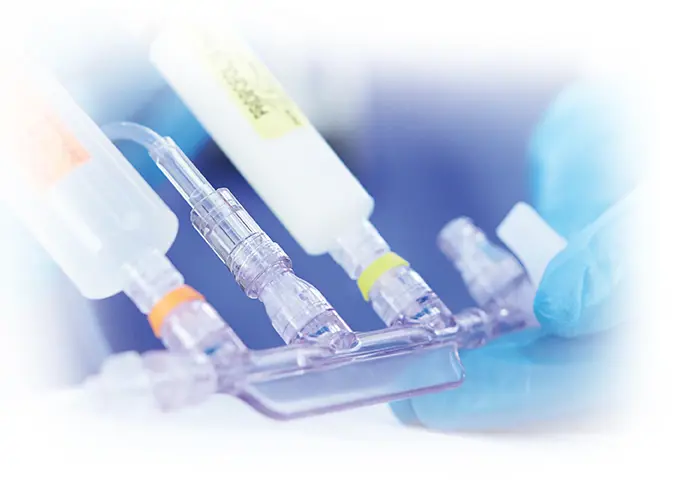
Using manifolds and stopcocks with Clave needlefree IV connector technology can help your efforts to minimize infection risks2
ICU Medical's full line of needlefree stopcocks and manifolds feature clinically differentiated Clave infection control technology designed to minimize the risk of contamination by maintaining a closed system. These access ports are ideal for anesthesiology, oncology, and critical care, where simultaneous fluid delivery is critical.
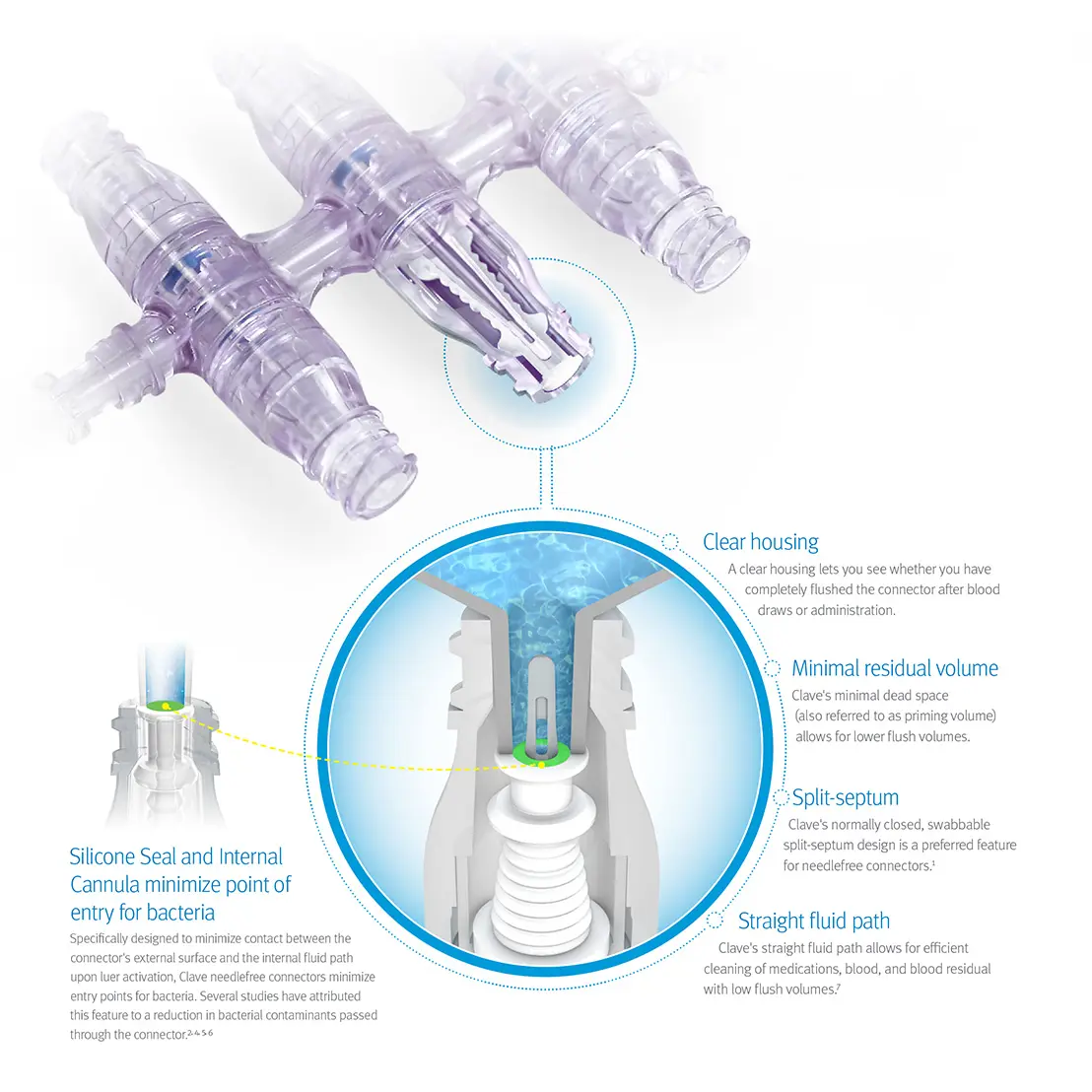
Clinically differentiated infection control technology
Clave needlefree IV connector technology can help your efforts to reduce infection risks by minimizing entry points for bacteria and maximizing the effectiveness of every flush, while helping you comply with CDC and Infusion Nurses Society (INS) guidelines.1,3
Enhance the safety of IV therapy in your specialty care areas
Clave technology helps enhance patient and clinician safety by using a single connector technology throughout the hospital so you can minimize clinical training and in-servicing while optimizing your supply chain.
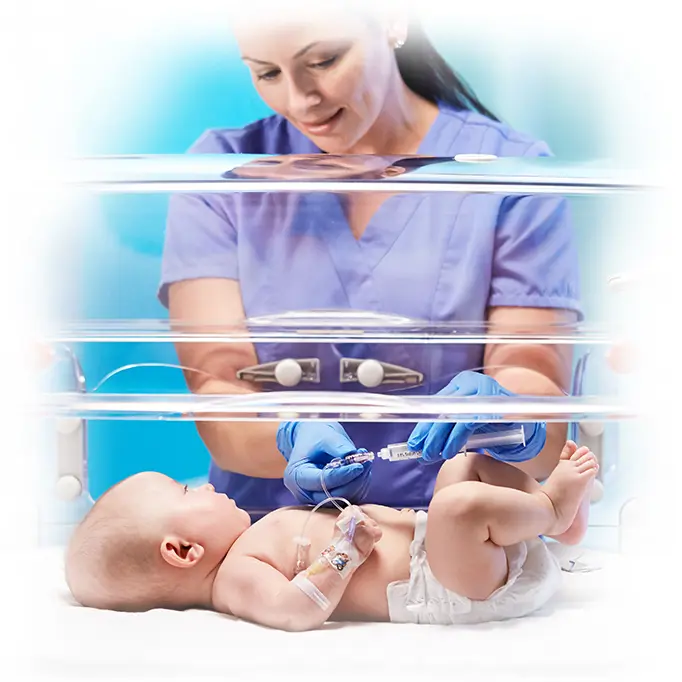
NICU and PICU
- Closed medication sets for syringe pump delivery
- Stopcocks and manifolds with low residual volumes
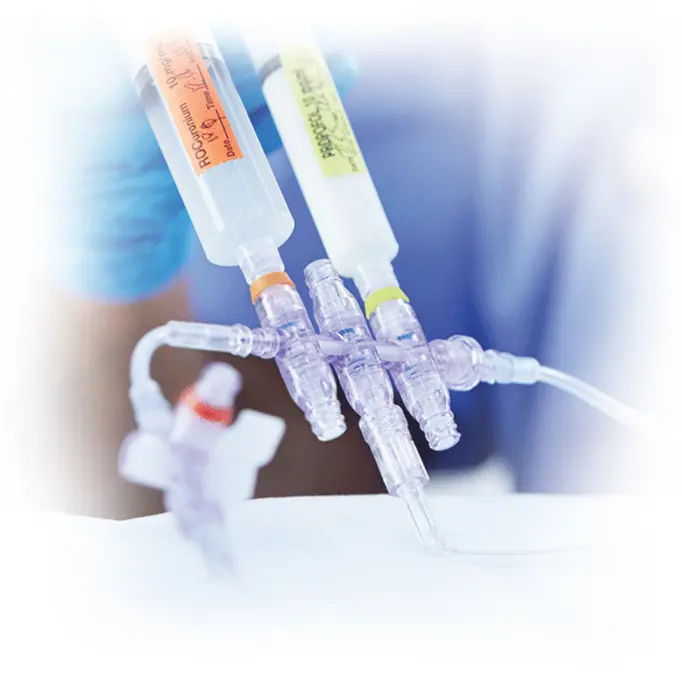
Anesthesia
- Multiport manifolds and stopcocks for simultaneous fluid delivery
- Custom administration, extension, and blood sets for adult and pediatric patients
Warning: Clave connectors may be incompatible with some male-luer connectors including prefilled glass syringes. To avoid damage to the Clave or syringes or male luers which may result in delays of medication administration and possible serious adverse events, users should confirm mating luers or syringes have an internal diameter range of 0.062” to 0.110”. Check the internal diameter of the male-luer connector of the mating syringe prior to using it to access the Clave. Products outside of these dimensional tolerances should not be used.
Product Information
Related Products
Product Inquiry
Please enter your details into the following form.
References
- Guidelines for the Prevention of Intravascular Catheter-Related Bloodstream Infections, Final Issue Review, May 17, 2010.
- Bouza E, Munoz P, Lopez-Rodriguez J, et al. A needleless closed system device (Clave™) protects from intravascular catheter tip and hub colonization: a prospective randomized study. J Hosp Infect. 2003; 54:279-287.
- Infusion Nurses Society (INS): Infusion Nursing Standards of Practice, 2011.
- Ryder, M., DeLancey-Pulcini, E., Parker, A., & James, G. (2023). Bacterial transfer and biofilm formation in needleless connectors in a clinically simulated in vitro catheter model. Infection Control & Hospital Epidemiology, 1-9. doi:10.1017/ice.2023.60
- JD Brown, HA Moss, TSJ Elliott. The potential for catheter microbial contamination from a needleless connector. J Hosp Infect. 1997.; 36:181-189.
- Yebenes J, Delgado M, Sauca G, Serra-Prat M, Solsona M, Almirall J, et al. Efficacy of three different valve systems of needlefree closed connectors in avoiding access of microorganisms to endovascular catheters after incorrect handling. Crit Care Med 2008;36: 2558–2561.
- Low Volume Flush Characteristics of Unique Needlefree Connectors M1-1223 Rev. 1.