SwabCap™
Disinfecting Cap for Needlefree Connectors
The only disinfecting cap to help enhance patient safety by providing the ability to disinfect and protect within 30 seconds for up to 7 days, if not removed1,2
- Continuous disinfection of connector surface
- Sterile, individual packaging
- Visual compliance confirmation
- Proprietary thread cover design
Maximize Infection Control Compliance with Easy-to-Use Disinfecting Technology
Nursing guidelines recommend swabbing needlefree connectors before every access to minimize the risk of bacterial contamination.3
Needlefree IV connectors play an important role in the fight against CRBSI, but nursing guidelines still suggest that connectors be swabbed before each access. Unfortunately, swabbing technique and compliance with these policies may vary, and visual confirmation of connector disinfection may be difficult.
SwabCap’s proven disinfecting technology can be an important element in your efforts to minimize infection risks and improve swabbing compliance.
SwabCap’s proprietary disinfecting cap design has been shown to help enhance the barrier to bacterial ingress while helping you standardize disinfection protocols.4 Unlike other caps that only disinfect upon application, SwabCap continues to disinfect the connector surface for up to 7 days until removed.
See How Something So Small is Becoming an Important Part of Infection Control
- The Infusion Therapy Standards of Practice (INS) recommends the use of passive disinfection caps containing 70% IPA, as they were associated with lower rates of CLABSI.
- The Society for Healthcare Epidemiology of Amerca (SHEA) recommends the use of disinfecting caps to help improve infection control best practices.5
- One study showed that use of SwabCap resulted in a 34% decrease in HA-CLABSI.6

Continuous
Disinfection
Completely disinfects after 30 seconds and continues disinfecting for up to 7 days, if not removed

Sterile,
Individual Packaging
Reduces the risk of cross contamination with individually packaged disinfecting caps

Visual Compliance
Confirmation
Helps ensure swabbing compliance with easily identifiable colored disinfecting caps

Proprietary Thread
Cover Design
Disinfects both the top and threads of the connector for maximum protection
Infection Control Technology Designed to Help Prevent Bacterial Contamination7
Its patented thread cover design gives SwabCap the unique ability to continue disinfecting both the connector’s surface and threads for up to seven days, if not removed
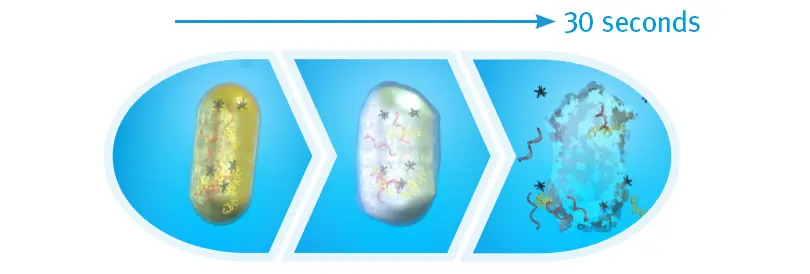
Bacterial Cell Death after 30 Seconds of IPA
When exposed to 70% isopropyl alcohol (IPA), harmful bacteria absorb the solution, making the cells swell, then breakdown and die. An in vitro study found that after 30 seonds of contact time with the cap, there were zero colony-forming units detected on the IV connectors.

Product Information
Related Products
Product Inquiry
Please enter your details into the following form.
References
-
Toxicon Thirty Second Disinfection Study for SwabCap, August 2018
-
Ethox International Seven Day Microbial Barrier Study for SwabCap, November 2008.
-
Infusion Nurses Society. Infusion nursing standards of practice. J Infus Nurs. 2021, 8th edition.
-
Posa P. Improving IV Connector Disinfection by Using Human Factors Engineering to Identify Effective, Nurse-Friendly Solutions. Poster presented at the APIC 4th Annual Conference. June, 2013.
-
Strategies to Prevent Central-Line Associated Bloodstream Infections in Acute Care Hospitals. Society for Healthcare Epidemiology of America (SHEA) 2014.
-
Kamboj M, Blair R, Bell N, et al. Use of disinfection cap to reduce central-line–associated bloodstream infection and blood culture contamination among hematology–oncology patients. Infect Control Hosp Epidemiol. December, 2015. 36:12.
-
Wright M, Tropp J, Schora D, et al. Continuous passive disinfection of catheter hubs prevents contamination and bloodstream infection. Am J Infect Control. 2012.